Background: The5 th (2022) edition of the WHO Classification for MDS recognizes MDS patients into two groups: MDS with defining genetic abnormalities and MDS morphologically defined. Further, the revised International Prognostic Scoring system (IPSS-R) assigns MDS patients into one of prognostic groups with distinct survival probabilities. However, both the IPSS-R and the 2022 WHO classification were developed based on data largely generated from patients in the high-income countries. There are limited data of how these systems perform in patients from developing and middle-income countries (LMIC). The primary objective of this study was to compare the performance of the two WHO classification: the revised 4 th (2016) and 5 th (2022) editions in IPSS-R defined Lower-Risk MDS Cohort of patients from LMIC in the GLAM registry.
Methods: TheGlam Registry enrolls patients from 16 Latin-American countries, For this analysis, we selected Lower risk MDS (defined as IPSS <3.5) patients from Argentina, Brazil, Chile, Colombia, Mexico, Paraguay, and Uruguay. Patients with CMML and higher-Risk MDS were excluded. The study was conducted in compliance with local regulations, and all subjects signed inform consents. Descriptive statistics, Sankey Diagram, Kaplan Meier methods and the Confidential Interval for the 5-year survival probability were used to report the results. Overall survival (OS) was measured from time of diagnosis to last contact or death, and progression-free survival (PFS) was measured from time of diagnosis to disease progression, progression to acute myeloid leukemia (AML), or death.
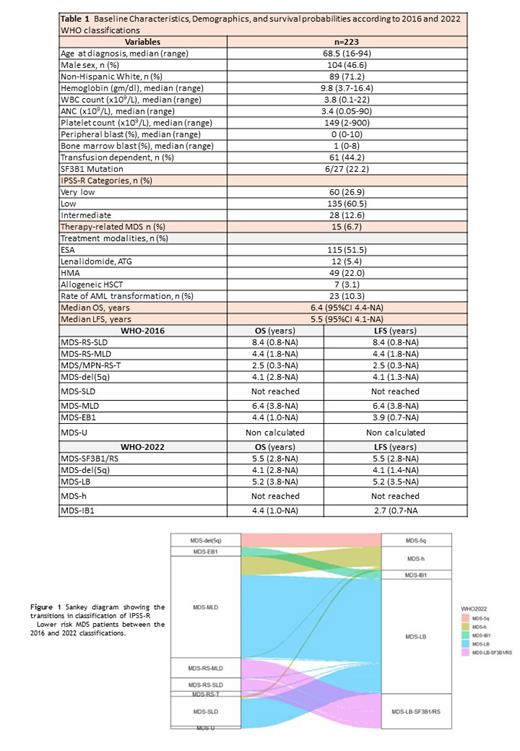
Results: A total of 223 LR-MDS patients were included in this analysis. Baseline characteristics and demographics are described in Table 1. Median age was 69 years, 47% were males, and 71% were non-Hispanic whites. The median blast count was 1% (range, 0-8%), and only 7% had therapy-related MDS. According to WHO-2016 edition, patients were classified as MDS-RS-SLD (n = 15 [6.72 %]); MDS-RS-MLD (n = 23 [10.3 %]); MDS-RS-T (n = 6 [2.7 %]), MDS-del(5q) (n = 15 [6.7 %]), MDS-SLD (n = 34 [15.2 %]), MDS-MLD (n = 116 [52 %]), MDS-EB1 (n = 11 [4.9 %]), MDS-EB2 (n = 0 [0 %]), and MDS-U (n = 3 [0.66 %]). According to WHO-2022 classification, subjects were classified as: MDS-del(5q) (n = 15 [6.7 %]), MDS-LB-SF3B1-RS (n = 40 [17.9% %]), MDS-biTP53 (n = 0 [0 %]), MDS-LB (n = 131 [58.7 %]), MDS-h (n = 27 [12.1 %]), MDS-IB1 (n = 10 [4.4 %]), MDS-IB2 (n = 0 [0 %]) and MDS-f (n = 0). Figure 1 represents the shifts in classification of patients between the 2016 and 2022 version. The 5-year survival probabilities (%) of MDS-SLD vs MDS-MLD (WHO-2016) was 62.5% (95CI 37.8-79.7) vs. 54.6% (95CI 39.9-65.6), and the 5-year PFS probabilities (%) for MDS-SLD vs MDS-MLD were 62.5% (95CI 37.8-79.7) vs 53.6% (95CI 39.9-65.6) respectively. There were no cases of biTP53-mutation among the 12.1% of patients who had testing for TP53. Three patients were re-classified from MDS-RS-MLD (WHO-2016) to MDS-LB (WHO-2022) because SF3B1 mutation was associated with complex karyotype, del(5q), del(7q) and/or TP53 monoallelic. Four patients with MDS-U (WHO-2016) were re-classified to MDS-LB (WHO-2022). MDS-LB category (WHO-2022) was a large and a very heterogeneous group with an OS and LFS of 5.2 years.
Conclusions: Our study, to our knowledge, provides one of the first, if not the first, datasets from LMIC to describe characteristics of IPSS-R lower-risk MDS pts and their re-classification and corresponding survival according to the WHO 2016 and 2022 classifications. Limited availability of molecular analysis in real-life settings (e.g., TP53 mutations) highlights some of the challenges of using the 2022 WHO classification (as well as IPSS-M) LMIC. Understanding the epidemiology of MDS pts in LMIC is important especially as some of the newly approved agents for lower risk MDS are starting to be used in these countries.
Disclosures
Zeidan:Chiesi: Consultancy, Honoraria; Gilead: Consultancy, Honoraria; Foran: Consultancy, Research Funding; Orum: Consultancy, Honoraria; Epizyme: Consultancy, Honoraria; Kura: Consultancy, Honoraria; Taiho: Consultancy, Honoraria; Tyme: Consultancy, Honoraria; Seattle Genetics: Consultancy, Honoraria; Mendus: Consultancy, Honoraria; Servier: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Daiichi Sankyo: Consultancy, Honoraria; Syros: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Ionis: Consultancy, Honoraria; Geron: Consultancy, Honoraria; Genentech: Consultancy, Honoraria; BeyondSpring: Consultancy, Honoraria; Boehringer-Ingelheim: Consultancy, Honoraria; Jazz: Consultancy, Honoraria; Agios: Consultancy, Honoraria; Regeneron: Consultancy, Honoraria; Shattuck Labs: Research Funding; Incyte: Consultancy, Honoraria; ALX Oncology: Consultancy, Honoraria; Celgene/BMS: Consultancy, Honoraria; Astellas: Consultancy, Honoraria; AbbVie: Consultancy, Honoraria; Schrödinger: Consultancy, Honoraria; Zentalis: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Notable: Consultancy, Honoraria; Lox Oncology: Consultancy, Honoraria; Otsuka: Consultancy, Honoraria; Syndax: Consultancy, Honoraria; Astex: Research Funding; Novartis: Consultancy, Honoraria; BioCryst: Consultancy, Honoraria. Apodaca Chavez:Astra Zeneca: Speakers Bureau. Gomez-De Leon:Abbvie: Honoraria; Astellas: Honoraria; AMGEN: Honoraria; Novartis: Honoraria; Jnssen: Other: Advisory board; Sanofi: Honoraria. Serrano:Bristol: Honoraria; Jansen: Honoraria. Grille:Abbvie: Speakers Bureau.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal